Immunohistochemistry is a key tool for the analysis of the localization of target molecules within tissues and is used routinely for almost every aspect of modern biomedical research. However, accurate results require an antibody that can distinguish between the intended molecule and related compounds.7 Antibodies are proteins in the immunoglobulin family that are produced by B-cell lymphocytes as a part of the adaptive immune response.8
Immunocytochemistry takes advantage of three properties of antibodies:
1. Antibodies uniquely bind to a protein or other molecule.
2. Antibody binding to molecules is essentially permanent in physiological conditions.
3. New antibodies can be made tailored to new interesting molecules. 9
ANTIBODY MOLECULES
When a molecule is present in a vertebrate animal that is not part of the “self,” it is processed by antigen-presenting T cells. These present the antigen to B cells, which bind it, and the B cells are activated to produce and secrete their own immune globulins. Immunoglobulins can be in several classes (IgG, IgM, IgA, IgE), which are produced at specific times and locations. 8
Figure.1 The antibody. An IgG antibody has a single constant region (white) with the Fc portion and the species-specific antigens. The variable region (gray) contains the Fab portion that binds the epitope portion of the antigen. The small protein, only in the variable region, is known as the light chain; the large protein that is part of the constant and variable region is the heavy chain. The IgG can be digested by the protease enzyme, papain, into an Fc end (constant end) and a Fab end (variable end).9
Immunocytochemistry uses antibodies against IgGs. The variable end of the antibody contains the unique epitope-binding regions that give each antibody its specificity (Figure 1. grey end). This variable region is the fraction antigen binding (Fab) portion. The unique configuration of the Fab specifically binds the epitope.
An antigen is a protein, peptide, or molecule used to cause an immune response in an animal. The animal responds by making antibodies to individual epitopes located on the antigen. An individual antigen has multiple epitopes that can generate antibodies.
An epitope can be an amino acid sequence on a denatured peptide or several sequences on the surface of a folded protein. 9
MAKING ANTIBODIES
The kinetics of antibody synthesis in the animal is of an apparently autocatalytic type, to a large number of interpretations. The theories of antibody formation can be divided into two classes, depending upon the assumption they make regarding the range of the antibody-forming capacity of a single cell: (a) restrictive theories, which postulate either one or at most, two antibody-forming potentialities in a single cell and (b) unrestrictive theories, which do not put any restriction to the synthetic capacity of a given cell.10
POLYCLONAL ANTIBODIES
An animal injected with an antigen will generate multiple antibodies to many epitopes. Antibodies are produced by B-cells and a single clone of B-cells produces antibodies to only a single epitope. Once a B-cell begins producing a single type of antibody, it will divide and give rise to many B-cells, all producing that single antibody to just one epitope; this is called a B-cell clone. Sometimes there are multiple clones of B-cells that produce antibodies to a single epitope. Parts of injected proteins and molecules make better antigens than others. As a result, some proteins do not generate many antibodies. 9
Figure 2. Polyclonal antibodies. An animal injected with an antigen will generate B-cell clones that can produce antibodies to multiple epitopes. The serum from the animal has different antibodies to these multiple clones, thus the name, polyclonal.9
Polyclonal antibodies contain multiple clones of antibodies produced to different epitopes on the antigen.
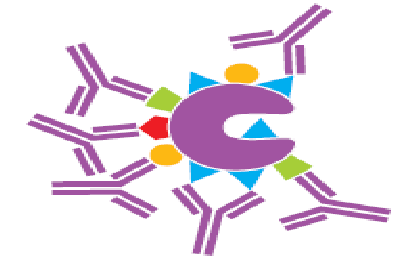
Figure 3. Polyclonal antibody binding to various epitopes.11
Advantages of Polyclonal Antibody
• Multiple clones give high levels of labeling for a single antigen because they contain many antibodies to different epitopes on the same protein.
Disadvantages of Polyclonal Antibody
• Shared epitopes on different proteins can label multiple proteins that are not antigen proteins.
• Obtaining the antibody depends on a living animal and the ultimate death of the rabbit means no more antibodies.
• When a new rabbit is immunized with the same antigen, the exact epitopes generating antibodies will be different and a different number of clones are generated. 9
MONOCLONAL ANTIBODIES
Monoclonal antibodies (originally from one mouse), contain a single antibody from one clone of B-cells to a single epitope on the antigen. This procedure was first described by Georges Kohler and Cesar Milstein, for which they received the Nobel Prize in 1984. Monoclonal antibodies are made by immunizing a mouse, and when antibodies are produced, the spleen of the immunized mouse is removed. The spleen cells are dissociated including the B-cells producing antibodies (Figure 4., different grey levels). Because B-cells will not divide in culture, they must be fused with a continuously dividing cell line that produces antibodies. Such a cell line is the mouse myeloma cell line. The spleen cells are fused with mouse myeloma cells to become a continuous hybridoma cell line. 9
Figure 4. Monoclonal antibodies. After injecting the antigen and generating several clones of antibodies, the spleen containing B-cells is removed. Hybridoma cells are made by fusing spleen B-cells with a myeloma cell culture line. 9
Three criteria to select the concentration of a monoclonal antibody to be used in an immunohistochemical method for paraffin-embedded tissues:
1. The monoclonal antibody should be diluted enough to stain the cells from which the immunizing antigen was obtained while not staining unrelated tissues from unrelated
species.
2. Staining should be obtained with the specific antibody but not with control monoclonal antibodies of unrelated specificity used at the same working concentration.
3. The staining patterns of monoclonal antibodies can be compared only if they are obtained from the same source, such as ascites or hybridoma supernatants. 12
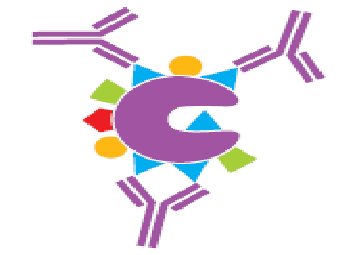
Figure 5. Monoclonal antibody reacts with specific epitope on an antigen.11
Advantages of Monoclonal Antibodies
• Single clone monoclonal antibodies bind to a single epitope, which is selected for high specificity for the antigen.
• Different clones of antibodies can be generated to different epitopes on a single antigen.
• Single clone can be generated to a post-transcriptionally altered protein (e.g., phosphorylated amino acid).
• Clones to an epitope shared with multiple proteins (gene products) can be rejected.
• The same antibody can be generated indefinitely from cultured hybridoma cells in a process that creates a stable reagent.
• The identical clones sold by different companies will be the same antibody.
Disadvantages of Monoclonal Antibodies
• Much work is required to generate a successful monoclonal antibody, especially in the cloning and selection process.
• Low levels of labeling occur because the monoclonal antibody binds an infrequent epitope on a protein or binds with low affinity.
• Monoclonal antibodies are mostly from mice because of a strong myeloma cell line.9
FINDING AND GETTING ANTIBODIES
Selecting an antibody can be a daunting task. Successfully using an antibody requires information about that antibody.
Catalog information – The catalog number and the price.
Description or background – The name of the antigen, its molecular weight, alternative names, and something about the antigen’s function.
Antibody type or host – The name of the species used to generate the antibody, the isotype of the antibody, and the clone name/number, if the antibody is monoclonal.
Source of antigen – The nature of the injected antigen (protein, peptide from a specific sequence) and the species of the antigen.
Packaging, product, or purification – The amount of the liquid product, the concentration of antibody in the product (1 mg/ml is ideal), additives (e.g., sodium azide, glycerol), the source (e.g., whole serum, supernatant, ascites), and purification, if any.
Specificity – A description of how the producer determined that the antibody binds only the listed antigen.9
Uses or application – The methods where the antibody has been tried are generally any of the following: immunohistochemistry (IHC), immunocytochemistry (ICC), or immunofluorescence (IF); western blot (WB) or immunoblot (IB); and immunoprecipitation (IP). This information should also include recommended dilutions for the listed applications.
Species reactivity – The species where the antibody binds the antigen. This is important because not all antibodies will bind equally to antigens from different species.
Protocols – The recommended protocol that can be useful to understand any unique fixation, detergent, blocking, or incubation conditions. 9
ANTIBODIES HANDLING AND STORING
Recommended Storage Freezer, –20◦C
• Repeated freezing and thawing will denature antibodies. Damage is reduced by diluting in 30% glycerol.
• Aliquot antibodies so they will be thawed once.
• Passive or non frost-free freezers at –20◦C or freezers at –70◦C. Not recommended are frost-free freezers that have circulating air which will dehydrate frozen antibodies in months.
Recommended Storage Refrigerator, 4◦C
• Prevents damage due to freeze–thaw.
• Non-frost-free refrigerators. Frost-free refrigerators will dehydrate and concentrate and eventually dry the antibody solutions.
• Add 0.02% sodium azide to inhibit the growth of bacteria. Note: sodium azide will inhibit an enzyme used for immunocytochemistry, horseradish peroxidase (HRP).
References:
7. Michael Levin. A novel immunohistochemical method for evaluation of antibody specificity and detection of labile targets in biological tissue. J. Biochem. Biophys. Methods 58 (2004) 85–96.
8. Clifford B. Saper. A Guide to the Perplexed on the Specificity of Antibodies. J Histochem Cytochem 2009 57: 1
9. R.W. Burry, Immunocytochemistry. Springer (2010)
10. Giuseppe Attardi, Melvin Cohn, Kengo horibata and Edwin S. Lennox. Symposium on the biology of cells modified by viruses or antigens. On the analysis of antibody synthesis at the cellular level. Vol 23.
11. George L Kumar and Lars Rudbeck. Immunohistochemical (IHC) Staining Methods, 5th Edition. Dako (2009).
12. D R Ciocca, D J Adams, R J Bjercke, G W Sledge, Jr, D P Edwards, G C Chamness, and W L McGuire. Monoclonal antibody storage conditions, and concentration effects on immunohistochemical specificity. J Histochem Cytochem 1983 31: 69.